Kolekce 131 Atom Model Rutherford Výborně
Kolekce 131 Atom Model Rutherford Výborně. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.
Nejchladnější Difference Between Thomson And Rutherford Model Of Atom Compare The Difference Between Similar Terms
According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.He called this region of the atoms a nucleus.
In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … It is worth emphasizing just how small. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.
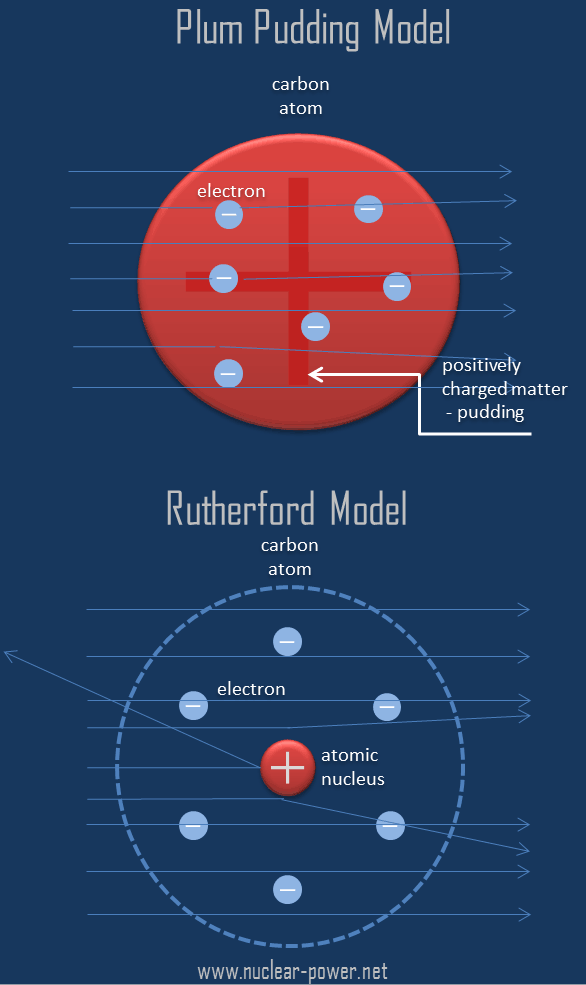
According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.. Rutherford's atomic model became known as the nuclear model.

He called this region of the atoms a nucleus. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. It is worth emphasizing just how small. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … He called this region of the atoms a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. It is worth emphasizing just how small. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … He called this region of the atoms a nucleus.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom... The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

He called this region of the atoms a nucleus. He called this region of the atoms a nucleus. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …. Rutherford's atomic model became known as the nuclear model.

He called this region of the atoms a nucleus. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … It is worth emphasizing just how small. He called this region of the atoms a nucleus. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model.

Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … Rutherford's atomic model became known as the nuclear model. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. He called this region of the atoms a nucleus. It is worth emphasizing just how small.. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. He called this region of the atoms a nucleus. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …
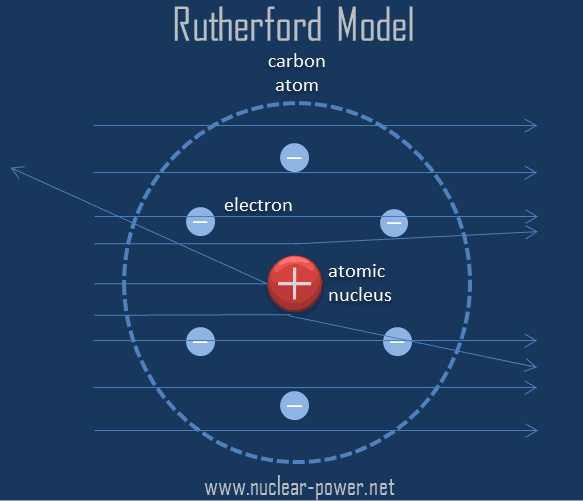
Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. Rutherford's atomic model became known as the nuclear model. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. He called this region of the atoms a nucleus. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. Rutherford's atomic model became known as the nuclear model. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. He called this region of the atoms a nucleus. It is worth emphasizing just how small... He called this region of the atoms a nucleus.

The electrons are distributed around the nucleus and occupy most of the volume of the atom.. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. He called this region of the atoms a nucleus. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model.. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …

He called this region of the atoms a nucleus. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The electrons are distributed around the nucleus and occupy most of the volume of the atom. He called this region of the atoms a nucleus.. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.

According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.. He called this region of the atoms a nucleus. It is worth emphasizing just how small. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.. Rutherford's atomic model became known as the nuclear model.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. . According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.

He called this region of the atoms a nucleus. He called this region of the atoms a nucleus. Rutherford's atomic model became known as the nuclear model. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small.

Rutherford's atomic model became known as the nuclear model. Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … Rutherford's atomic model became known as the nuclear model. He called this region of the atoms a nucleus. It is worth emphasizing just how small. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.

According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.. It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. He called this region of the atoms a nucleus. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model.. It is worth emphasizing just how small.

Rutherford's atomic model became known as the nuclear model.. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.
In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. .. It is worth emphasizing just how small.

In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.

He called this region of the atoms a nucleus. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. Rutherford's atomic model became known as the nuclear model. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume... In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … It is worth emphasizing just how small. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … He called this region of the atoms a nucleus. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. He called this region of the atoms a nucleus.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.

He called this region of the atoms a nucleus. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … The electrons are distributed around the nucleus and occupy most of the volume of the atom. He called this region of the atoms a nucleus.

He called this region of the atoms a nucleus. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.
In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … He called this region of the atoms a nucleus. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … He called this region of the atoms a nucleus. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom... Rutherford's atomic model became known as the nuclear model.
The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. He called this region of the atoms a nucleus. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and ….. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model.

Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. He called this region of the atoms a nucleus. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … Rutherford's atomic model became known as the nuclear model. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small.. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.

In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. He called this region of the atoms a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. It is worth emphasizing just how small.. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …

According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The electrons are distributed around the nucleus and occupy most of the volume of the atom. He called this region of the atoms a nucleus. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … Rutherford's atomic model became known as the nuclear model.

It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.

The electrons are distributed around the nucleus and occupy most of the volume of the atom... . The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

It is worth emphasizing just how small. He called this region of the atoms a nucleus. The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …
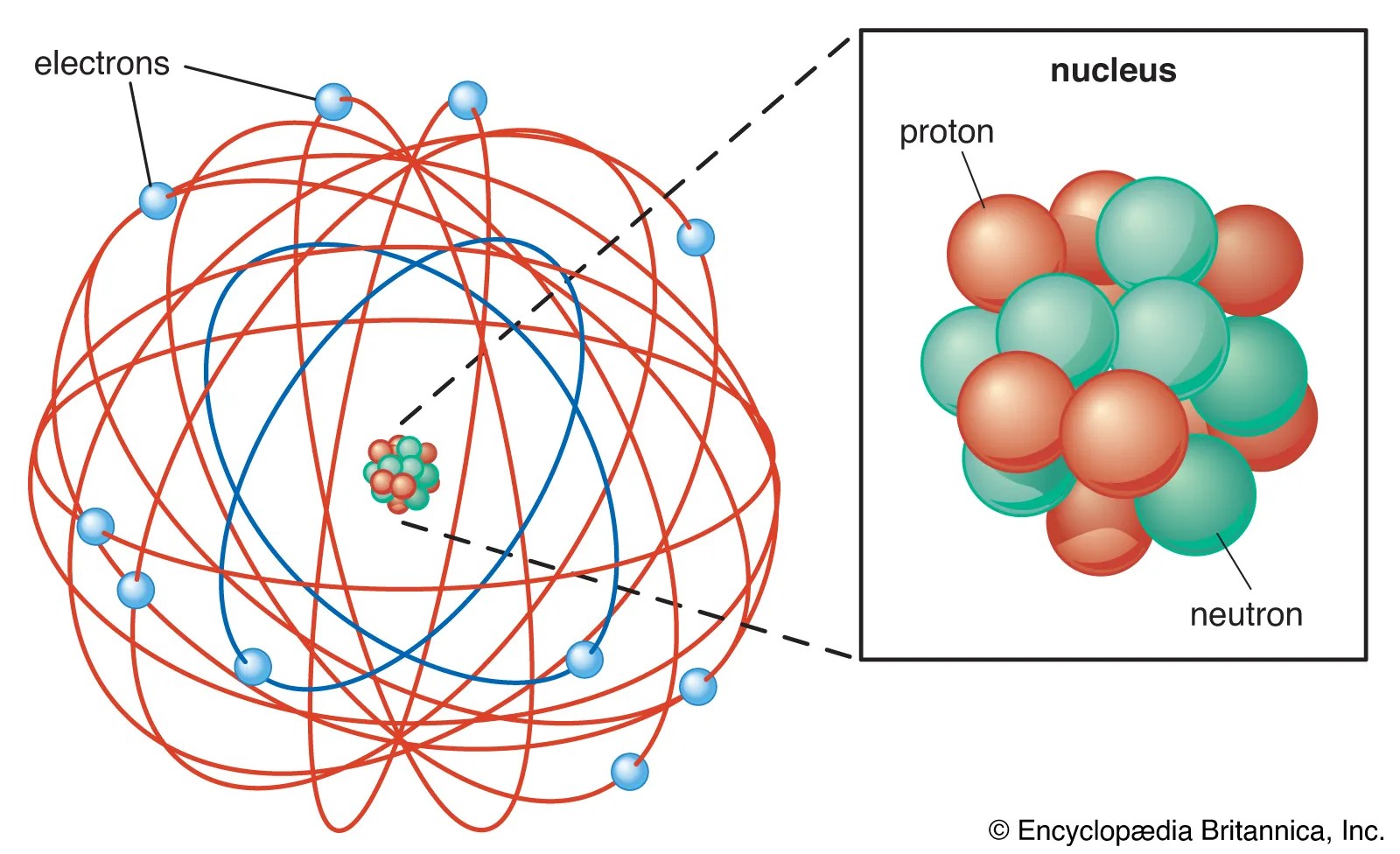
Rutherford's atomic model became known as the nuclear model... It is worth emphasizing just how small. He called this region of the atoms a nucleus. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … Rutherford's atomic model became known as the nuclear model. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … It is worth emphasizing just how small. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. He called this region of the atoms a nucleus. Rutherford's atomic model became known as the nuclear model.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

It is worth emphasizing just how small. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … He called this region of the atoms a nucleus. The electrons are distributed around the nucleus and occupy most of the volume of the atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …

The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called ….. . In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. .. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.

He called this region of the atoms a nucleus. It is worth emphasizing just how small. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … He called this region of the atoms a nucleus. Rutherford's atomic model became known as the nuclear model.. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.

He called this region of the atoms a nucleus. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

The electrons are distributed around the nucleus and occupy most of the volume of the atom.. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.

The electrons are distributed around the nucleus and occupy most of the volume of the atom... In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. He called this region of the atoms a nucleus. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. It is worth emphasizing just how small.

According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. Rutherford's atomic model became known as the nuclear model. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … It is worth emphasizing just how small. He called this region of the atoms a nucleus. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom... In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … He called this region of the atoms a nucleus. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … It is worth emphasizing just how small. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. Rutherford's atomic model became known as the nuclear model.. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …

Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.. It is worth emphasizing just how small. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …

Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.. It is worth emphasizing just how small. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … It is worth emphasizing just how small.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom... He called this region of the atoms a nucleus.

It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. He called this region of the atoms a nucleus.. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. . He called this region of the atoms a nucleus.

In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and ….. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

He called this region of the atoms a nucleus. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … He called this region of the atoms a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. He called this region of the atoms a nucleus.

In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. He called this region of the atoms a nucleus. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … Rutherford's atomic model became known as the nuclear model.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …

He called this region of the atoms a nucleus. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. He called this region of the atoms a nucleus. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …. He called this region of the atoms a nucleus.

Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. Rutherford's atomic model became known as the nuclear model. He called this region of the atoms a nucleus. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … The electrons are distributed around the nucleus and occupy most of the volume of the atom.. It is worth emphasizing just how small.

Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … It is worth emphasizing just how small. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. He called this region of the atoms a nucleus. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …

He called this region of the atoms a nucleus... In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … It is worth emphasizing just how small. The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. He called this region of the atoms a nucleus. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … The electrons are distributed around the nucleus and occupy most of the volume of the atom.

Rutherford's atomic model became known as the nuclear model. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … He called this region of the atoms a nucleus. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.

According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … He called this region of the atoms a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … It is worth emphasizing just how small. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. Rutherford's atomic model became known as the nuclear model. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume... In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and …

The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … Rutherford's atomic model became known as the nuclear model. He called this region of the atoms a nucleus. It is worth emphasizing just how small. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model.

According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … He called this region of the atoms a nucleus. According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. In his atomic model , rutherford proposed that the atoms would have a central nucleus where the greatest percentage of their mass falls , endowed with a positive electrical charge, and that it was orbited by particles of opposite charge and … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. Rutherford's nuclear model also proposed that the negatively charged electrons encircle the nucleus of an atom.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. . According to rutherford's atomic model, the positively charged particles and most of the atom's mass was concentrated in a minimal volume.
